Good News At Last! The Covid Vaccine is on its way
We are waking up this morning to news that the Medicines Regulatory Health Agency (MRHA) in the UK has approved the use of the BioNTech/Pfizer Covid-19 vaccine. I’m sure hot on its heels Moderna and Astra Zeneca will hopefully follow suit.
 Source: Al Jazeera
Source: Al Jazeera
Moderna and Pfizer-BioNTech (which showed >90% immune response) have been in a tight race to launch their COVID-19 vaccines in Europe after both applied for emergency EU approval on 1st December. The applications to the European Medicines Agency (EMA) came a day after Moderna sought emergency use for its vaccine in the United States and more than a week after Pfizer and BioNTech did the same.
So, this morning the Northern Ireland Health Minister Robin Swann hoped that they could start vaccinating healthcare workers and the elderly in the coming weeks. Let’s hope the EMA (the regulator for Ireland) approval comes soon too. Whilst fantastic news, this is just the beginning of the vaccination program. With the doses expected to arrive the estimates are that initially only 0.5% of the population will be vaccinated and they need two doses, so this takes time, and also allow time for the body to produce its immune response time too. So it doesn’t mean we can suddenly give up all the hard work of the last year keeping our social distance, it will take some months for enough of the population to be vaccinated for us to get back to some normality. But it offers hope, and that’s what we all need right now.
When the other companies get their approvals from the appropriate regulators and production of the vaccines are scaled up around the world, further sectors of the population will then be able to be vaccinated.
When can we expect the vaccine to roll out in Ireland?
Any clearance in the EU and United States will be “conditional” or for “emergency use”, respectively, meaning developers are obliged to continue trials and provide more trial results as they emerge. The European filing completes the so-called rolling review process, which was initiated with the EMA on 6 October.
If EMA concludes that the benefits of the vaccine outweigh its risks in protecting against COVID 19, it will recommend granting a Conditional Marketing Authorisation. The European Commission will then fast-track its decision-making process with a view to granting a CMA valid in all EU and EEA Member States within days, to enable the roll-out of the vaccination plan.
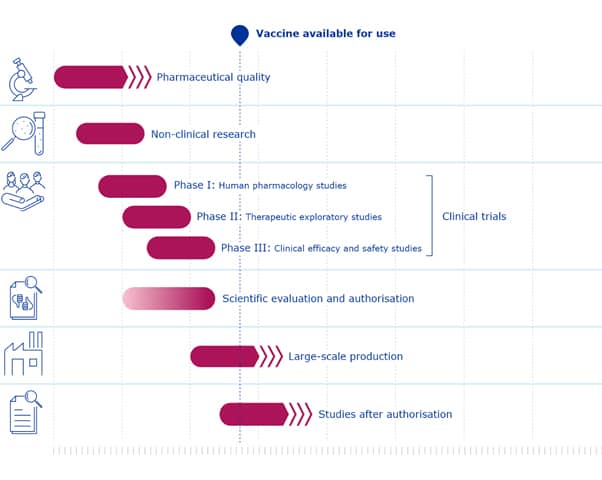
How has the Covid-19 vaccine happened so fast?
In the past, vaccines have been developed through a series of steps that can take many years. Phase One of human trials are usually carried out on 30 to 50 people to make sure the vaccine is safe and does not have any unexpected side effects. Phase Two involves a larger trial that begins to look at the vaccine’s immunogenicity, which is whether it elicits the required immune response. Phase Three trials usually involve tens of thousands of people and test the efficacy of the vaccine, which is how well it protects a person against infection, as well as its safety in such a large group.
Now, given the urgent need for a COVID-19 vaccine, unprecedented financial investments and scientific collaborations are changing how vaccines are developed. This means that some of the steps in the research and development process are happening in parallel, while still maintaining strict clinical and safety standards. For example, some clinical trials are evaluating multiple vaccines at the same time. However, this does not make the studies any less rigorous.
Will it be safe?
As for all medicines, EU authorities continuously collect and review new information on medicines once they are on the market and take action when needed. In line with the EU safety monitoring plan for COVID-19 vaccines, monitoring will take place more frequently and will include activities that apply specifically to COVID-19 vaccines. Companies for example will provide monthly safety reports in addition to the regular updates required by the legislation and conduct studies to monitor the safety and effectiveness of COVID-19 vaccines after their authorisation.
These measures will allow regulators to swiftly assess data emerging from a range of different sources and take appropriate regulatory action to protect public health if needed.
How do these vaccines work?
Both Moderna and Pfizer-BioNTech have reported preliminary findings of more than 90% effectiveness – an unexpectedly high rate – in trials of their vaccines, which are both based on new messenger RiboNucleicAcid (mRNA) technology.
Their work validates that of several biotech experts, who for years have been labouring to prove a once-unorthodox idea: The human body can act as its own vaccine factory.
Both inoculations are expected to work by preparing the body to defend itself against infection with the Covid-19 (coronavirus SARS-CoV-2). The virus uses a protein on its outer surface called a “spike protein” to enter the body’s cells and cause disease. The vaccine contains the genetic instructions (mRNA) to produce the spike protein. The mRNA is covered in small lipid (fat) particles that help deliver the mRNA into the cells and prevent it from being degraded. When a person is given the vaccine, their cells will read the genetic instructions and produce the spike protein. The person’s immune system will then treat this protein as foreign and produce natural defences — antibodies and T cells — against it. If, later on, the vaccinated person comes into contact with covid-19, the immune system will recognise the virus and be prepared to attack it: antibodies and T cells can work together to kill the virus, prevent its entry into the body’s cells and destroy infected cells, thus helping to protect against COVID-19.
It’s worthwhile pointing out that the WHO estimate that up to 150 vaccines using different technologies are still being developed and trialed around the world.
Time will tell how the other vaccines will develop and be approved. The AstraZeneca-Oxford vaccine has a practical advantage over some others in that it can be stored at 2-8°C (normal fridge) rather than – 70°C Celsius needed for the Pfizer and -20°C Moderna vaccines, for example.
What next?
Almost 4.4 billion doses of the various vaccines have been pre-ordered around the world, according to Reuters news agency. A fierce international competition to lock in billions of dollars-worth of orders for the vaccine has been under way for months, and some companies will start delivering millions of doses as soon as mid-December. As such, it will more likely be early summer before a large part of the world population is inoculated.
We wait for EU and US approvals for the vaccine and hope they’ll come soon to start the vaccine roll-out early in January.
The advice for now is still to observe your distance, wear face coverings and limit your contacts. But it’s great now to have something to look forward to for 2021!